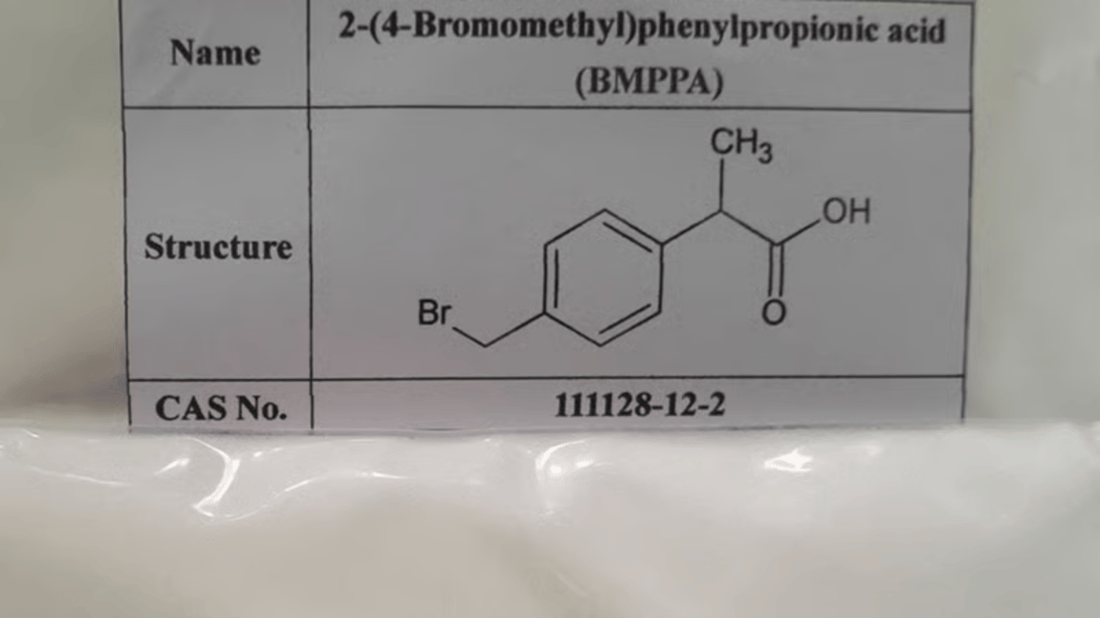
Chemical Structure and Properties of 2-(4-bromomethyl)phenylpropionic Acid
2-(4-bromomethyl)phenylpropionic acid (Cas No. 111128-12-2) is a brominated aromatic compound featuring a propionic acid moiety attached to a phenyl ring substituted with a bromomethyl group at the para position. The molecular formula C10H11BrO2 reflects the presence of bromine, a halogen that influences its reactivity and physicochemical characteristics. This compound exhibits distinct melting and boiling points, solubility profiles, and stability parameters critical for its handling and usage in chemical processes.
Synthetic Routes and Production Methods
The synthesis of 2-(4-bromomethyl)phenylpropionic acid primarily involves selective bromination and functional group transformations starting from phenylpropionic acid or similar precursors. Techniques often utilize brominating agents under controlled conditions to achieve para-substitution on the methyl position of the phenyl ring. Optimization of reaction parameters ensures high yield and purity, which is essential for industrial and research applications.
Applications in Pharmaceutical Intermediate Development
This compound serves as a valuable intermediate in the pharmaceutical industry. Its bromomethyl functional group acts as a reactive handle for further chemical modifications, enabling the synthesis of complex molecules such as anti-inflammatory agents or selective enzyme inhibitors. The compound’s ability to undergo nucleophilic substitution reactions facilitates the incorporation of diverse bioactive moieties.
Role in Organic Synthesis and Chemical Research
In organic synthesis, 2-(4-bromomethyl)phenylpropionic acid (Cas No. 111128-12-2) offers a unique scaffold for constructing substituted aromatic compounds. Researchers utilize it to explore reaction mechanisms, develop new catalysts, and modify molecular frameworks. Its bromomethyl group promotes regioselective reactions, making it a versatile reagent in advanced chemical methodologies.
Environmental and Safety Considerations
Handling of 2-(4-bromomethyl)phenylpropionic acid necessitates awareness of its potential hazards. As a brominated compound, it exhibits moderate toxicity and should be managed with appropriate protective equipment. Environmental exposure must be controlled to prevent contamination, as brominated organics can pose risks to aquatic life and ecosystem balance. Proper disposal and containment protocols are vital.
Analytical Techniques for Quality Control
Characterization and quality assurance of 2-(4-bromomethyl)phenylpropionic acid rely on robust analytical methods. Techniques such as nuclear magnetic resonance (NMR) spectroscopy, mass spectrometry (MS), and high-performance liquid chromatography (HPLC) are essential for confirming structural integrity and purity. Additionally, infrared (IR) spectroscopy aids in identifying functional groups, ensuring compliance with specification criteria.
Market Availability and Commercial Suppliers
Due to its specialized uses, 2-(4-bromomethyl)phenylpropionic acid (Cas No. 111128-12-2) is predominantly available through chemical suppliers focusing on research and pharmaceutical intermediates. Commercial availability varies based on purity grades, bulk quantities, and regional regulations. Pricing and lead times fluctuate depending on demand and synthesis complexity, making early procurement planning advisable.
Regulatory Framework and Compliance
The production, sale, and use of 2-(4-bromomethyl)phenylpropionic acid are subject to chemical safety regulations in many countries. Material Safety Data Sheets (MSDS) provide guidance on handling and emergency procedures. Compliance with local and international chemical control regulations ensures safe usage and legal procurement, particularly in pharmaceutical manufacturing and laboratory research.
Innovative Research and Derivative Development
Recent scientific studies explore modifications of 2-(4-bromomethyl)phenylpropionic acid to develop novel derivatives with enhanced biological activity. Such research aims at creating targeted drug candidates or specialized materials with unique properties. Functionalization strategies leverage the bromomethyl substitution to introduce diverse chemical groups, broadening the compound's applicability.
Future Prospects and Industrial Significance
The future potential of 2-(4-bromomethyl)phenylpropionic acid (Cas No. 111128-12-2) lies in its role as a building block for advanced pharmaceuticals and specialty chemicals. Ongoing innovation in green synthesis methods and scalable production techniques promises to enhance its commercial viability. Increasing demand for tailored intermediates in drug discovery underscores its importance in modern chemical industries.
Quote Inquiry
contact us